Whether for pharma, fine chemical, agrochemical, electronic or natural product research, in recent years, there has been increasing pressure to achieve and deliver higher purity products. Chemical products derived from various synthetic chemistry approaches carry with them an inherent risk of by-products and therefore contamination can be a big problem. Potential contaminants are numerous and can be solvent, reagent or catalyst derived...
In recent years, there has been great focus and emphasis on greener solutions for chemistry. The use of atom efficient and environmentally friendly catalysts has increased, resulting in the explosion in use of transition metal catalysts. One of the most common synthetic transformations in this respect is the creation of the C-C bond. Advances in catalysis research, leading to more efficient transition metal reactions have led the most successful proponents of this chemistry to the highest acclaims. One such reaction is the Suzuki-Miyaura1 reaction, which is now one of the most commonly employed C-C bond forming reactions in chemistry, using Palladium catalysts.
Being highly effective and used in trace amounts is a double-edged sword because those metals can be difficult to remove from the products that they helping to create, after the chemical reaction. There is a plethora of classical techniques such as distillation, recrystallization, carbon adsorption etc. that can be used to purify products such as APIs, however in recent years, regulation has tightened and achieving purity levels for metals has become increasingly challenging for those classical techniques. Some existing processes for example now fail to output product of the required purity standards, and so the industry has had a major issue on its hands.
In our work, we investigated the use and application of solid supported metal scavengers. These are completely insoluble adsorbents that may be added to reactions, which bind metal and can be removed easily afterwards. To look at this in greater detail, we initially focused our attention on a powerful scavenger for palladium, called Biotage® MP-TMT.
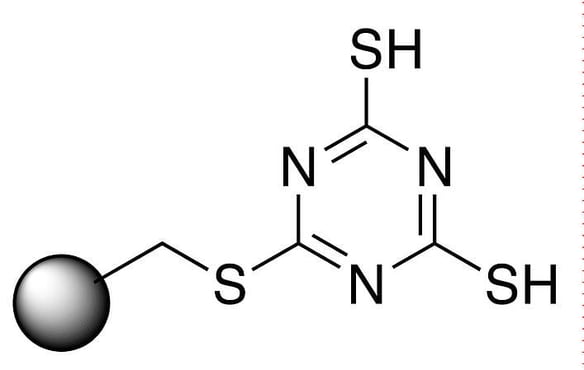
Figure 1 – MP-TMT
Procedures for using metal scavengers are relatively simple. Biotage® MP-TMT (Figure 1) is a macroporous cross-linked polystyrene / divinylbenzene polymer functionalized with a dimercaptotriazinethiomethyl group. We add a defined amount to a reaction, stir and filter. Since our experiment was a batch stir process, equilibration was not necessary. To check results we used a third party independent UKAS accredited (to ISO 17025:2017) lab for ICP to give an accurate representation of the palladium content of our samples.
Our results, using a classical Suzuki reaction as a probe, coupling an aryl-bromide with a boronic acid are shown below (Figure 2).
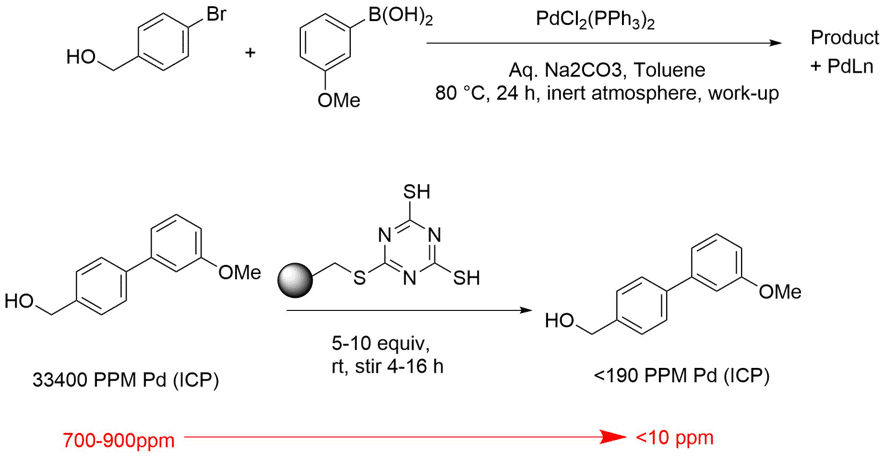 Figure 2 - Chemical reaction probe to assess metal scavengers
Figure 2 - Chemical reaction probe to assess metal scavengers
Chemistry wise, of course this was going to work, however we didn’t know the impact and resulting distribution of the palladium from the catalyst. Using a very highly loaded catalytic system, we obtained our crude product, conducted the typical work-up, washed with brine and extracted our product into the organic to isolate organic bi-aryl coupled product. Using elemental analysis, we measured levels of palladium before and after a scavenging protocol, in which we simply added 5 equivalents of the MP-TMT metal scavenger to the reaction (with respect to palladium) and stirred the reaction at RT overnight. Our heavily loaded system started out at 33,000ppm, and after one treatment, we saw that Pd levels had been reduced to less than 200ppm.
Repeating the experiment using the more typical 0.5 wt % of the palladium catalyst (we used about 2% initially in our heavily loaded stress test), we have consistently seen resulting palladium levels pre-metal scavenging treatment of 700-900ppm and post treatment, at under 10ppm.
In conclusion, we added MP-TMT in stoichiometric quantities to the palladium used in our Suzuki reaction (5eq) which reduced the metal contamination in our final product by a factor of between 100x and 1000x. We isolated sufficiently pure product with less than 10ppm Pd, from a simple overnight stir treatment.
We have further optimized our scavenging protocols for both time, and equivalency, please click here for more information from our metal scavenging microsite.
1) Miyaura, Norio; Yamada, Kinji; Suzuki, Akira (1979). A new stereospecific cross-coupling by the palladium-catalyzed reaction of 1-alkenylboranes with 1-alkenyl or 1-alkynyl halides. Tetrahedron Letters. 20 (36): 3437–3440.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership