It’s engrained in our minds and psyche, from earliest school days and exposures to science that heating a reaction speeds it up. Later, we learn some more of the details, that by increasing the temperature of a reaction, we can double its reaction rate. And after, the Arrhenius equation (Figure 1) is revealed to us, completing the picture and making the science infallible.
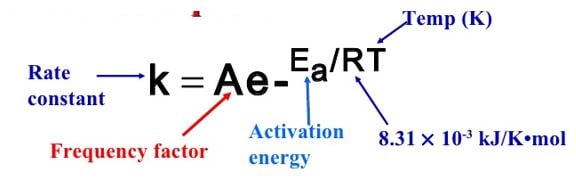
Figure 1. – the Arrhenius equation
But the implications of what happens in more complex chemical situations can be slightly less intuitive and so I thought I would briefly look into this after receiving questions during some of my seminars.
So…we know, heating a reaction will increase its reaction rate. But what about heating a metal scavenging reaction? The paradigm shift here is that we have two completing reactions occurring. Since metal scavenging can be described as follows (Figure 2).....
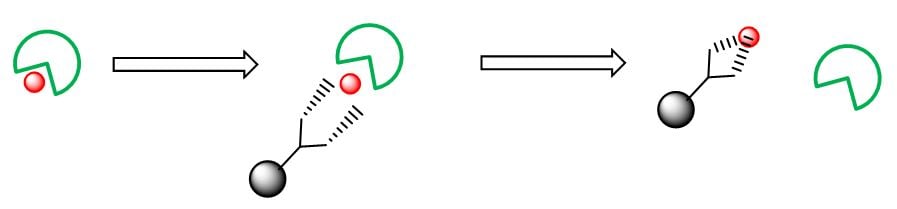
Figure 2. – conceptual view of metal scavenging
….we need to both break the bond between the metal (red dot) and the chemical product (green wedge) and then form a bond between the metal scavenger and the metal (right hand side). So, heating will help to break the first set of bonds and complete part one of the process but it can also destabilize the formation of the ‘scavenger to metal complex’ by raising the overall energy levels (of the final product) in comparison to it’s forward activation energy. A double-edged sword, well that’s the theory at least.
So we looked at two powerful industry established metal scavengers for palladium as we have done before in our blog series but this time we focused on 2 different scavenging mechanisms, ionic vs transition metal : ligand binding with Biotage® MP-TMT, ISOLUTE® SCX-2 (Figure 3)
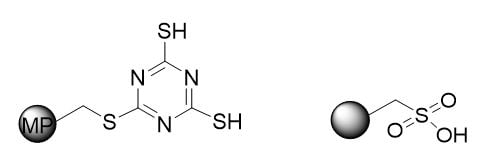
Figure 3. – Structure of Biotage MP-TMT (left) and ISOLUTE® SCX-2 metal scavengers
The style of reaction was similar to a pattern we have used before, batch scavenging (2 hr stir) of a 1000ppm Dichlorobis triphenylphosphine Pd II catalyst at 3 different temperatures (i)200C (ii) >400C (iii)<300C
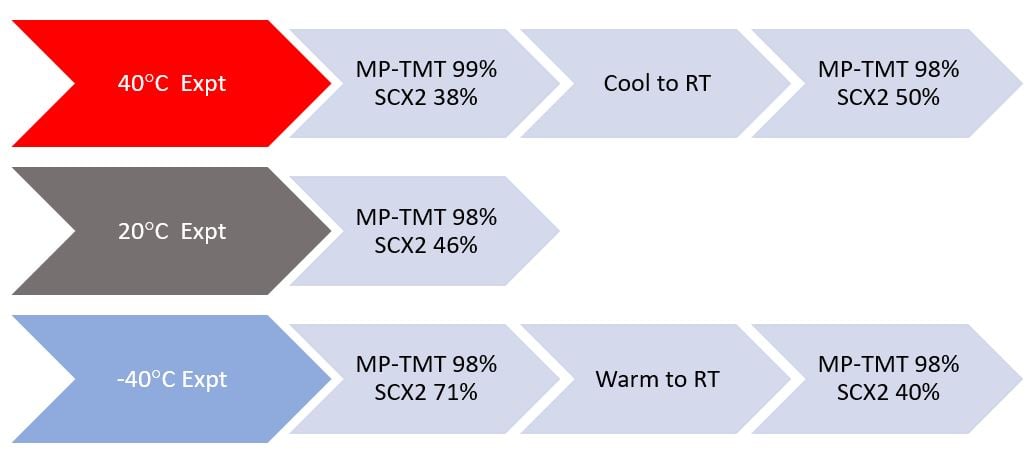
Figure 4. shows results from metal scavenging reactions conducted at different temperatures
There are 3 swim lanes in Figure 4, corresponding to scavenging at each of 3 different laboratory set reaction temperatures. Since we did not want to re-invent palladium chemistry, we just used a convenient pre-made 1000ppm Pd stock solution of catalyst as our target.
At room temperature, Biotage MP-TMT and SCX-2 removed Pd in the amount of 98% and 46%. These two scavengers use different mechanisms, and in this system, we knew there would be differences. (SCX-2 is not normally optimized for Pd scavenging in this way btw, so 46% isn’t ‘bad’ per say).
Scavenging at 400C, barely increased the efficiency of scavenging with MP-TMT (to 99%), as it was already very efficient but the 400C reaction temperature had a negative impact on the other mechanism, that of ion-exchange type metal scavenging that could be expected with SCX-2.
Metal scavenging at -400C again had negligible impact on the MP-TMT scavenging reaction, but significantly increased the efficiency of the SCX-2 system. This gave an interesting insight to the effect of temperature and the relative bond strengths formed by metal scavengers vs the chemical binding mechanisms in play.
MP-TMT has an opportunity to form a very strong bidentate bond with Pd, whereas SCX-2 is more interested in ionic interactions under those conditions. (Clearly pH is going to be a further factor here, but for this experiment, we used mild neutral conditions)
The really interesting part to the confirmation of our postulation above came when we ran the first part of the reaction with the metal scavengers again but then adjusted the final set-hold temperature of the scavenging systems. In the case of SCX-2 metal removal wasn’t really favored at 400C (38% completion), but did increase to 50% completion on cooling to RT. When chilled to -400C SCX-2 was most active but lost its activity and returned to an efficiency of ~40% on warming to RT. This was similar to the straight-out RT scavenger reaction datapoint (@46%). [Note this also speaks volumes about the reversibility of this type of reaction, but I’ll save that for another blog]
So, temperature can destabilize ionic scavenging interactions. In the case of MP-TMT, there was little impact from temperature suggesting the bonds that form when that scavenger gets to work are both strong and relatively irreversible under these conditions. This means from a scale up perspective, we must also consider process economics. There is little point utilizing additional energy to heat or cool a system to improve scavenging if the improvement will only be marginal.
There are several complicating caveats of course, so we can’t make absolute rules out of this. If a sample is viscous for example, then heating it will loosen it up to a point that scavenging efficiency is increased – we always have to think about the mass transport and concentration. And if a metal scavenging mechanism is likely to have a dominant ionic / ion pair contribution, it could be helpful to consider cooling the reaction (but we’ve not seen many cases where this has been necessary).
So when we can categorize our metals removal process to be ‘normal’, it’s best to apply metal scavengers at room temperature to get the best of both worlds, efficiency, and energy utilization.
Read our other blog posts to learn more about metal scavenging, just follow the link below!

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership