Cannabis entrepreneurs continually seek to differentiate themselves from others in the market. Some focus on THC while others focus on CBD.What I have seen recently after attending some cannabis-specific conferences is a growing interest in isolating/purifying some of the minor, naturally occurring phytocannabinoids such as CBG and CBDV.
In this post, I discuss some work I recently performed in my lab with a hemp extract where I separated, identified, and isolated CBG and CBDV.
For those needing to purify cannabis and hemp extracts in order to remove impurities and separate/isolate individual cannabinoids, one of the most successful tools used to achieve these goals is flash chromatography. For those unfamiliar with flash chromatography, it is a process that uses an automated purification system consisting of a pump, a column filled with media (either a waxy sorbent or silica), solvents (ethanol + water, or hexane + ethyl acetate), an ultraviolet detector (detects the cannabinoids as they exit the column), and a fraction collector (collects the detected compounds) – all controlled by software.
There are two useful flash chromatography modes, normal-phase and reversed-phase. Normal-phase uses a silica filled column and non-polar solvents such as hexane and ethyl acetate while reversed-phases uses a waxy, bonded silica with water and ethanol as solvents. Their separation mechanisms are different and your purification goals are will dictate which mode to use.
For instance, to remediate THC from CBD, reversed-phase flash chromatography is the best mode because it separates compounds with different hydrophobicities, Figure 1.
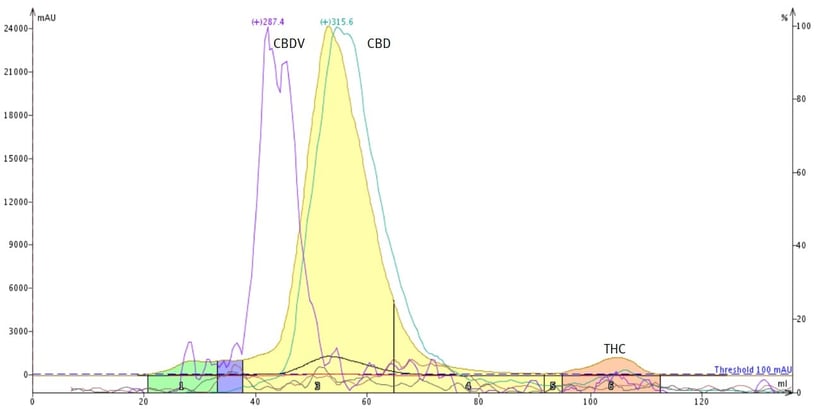
Figure 1. Reversed-phase flash chromatography removes THC from CBD and partially removes CDBV.
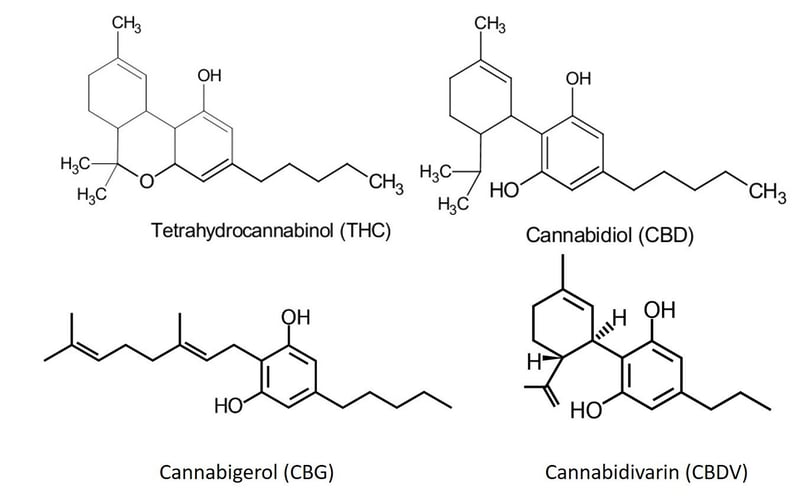
Figure 2. Chemical structures of THC, CBD, CBDV, and CBG account for the differences in polarity and hydrophobicity.
CBD differs from THC only in the number of -OH functional groups, CBD has two while THC contains only one which impacts hydrophobicity more than polarity. CBG has two -OH groups like CBD but lacks a second six-membered ring which impacts polarity. CBDV has fewer carbon atoms than the others impacting hydrophobicity.
What about CBG and THCV? Can the structure differences be exploited using flash chromatography? If so, which mode works best for them?
Well, we know that we can separate THC, CBD, and CBDV using reversed-phase because of hydrophobicity differences. The separation of CBD and CBG, however, using reversed-phase flash chromatography is difficult because the hydrophobicity differences are not large enough to exploit, resulting in co-elution, Figure 3.
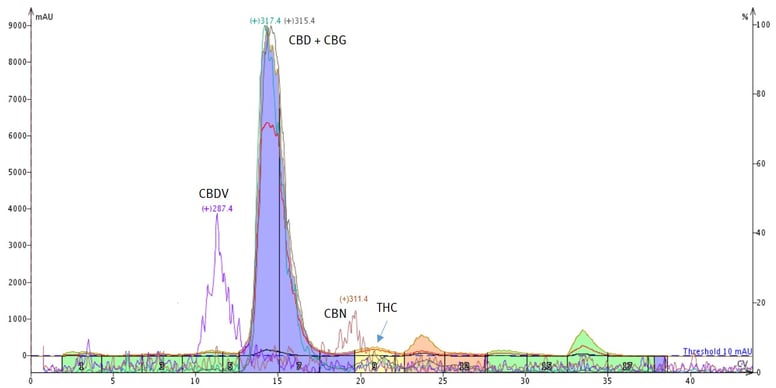
Figure 3. Reversed-phase hemp distillate purification shows the separation of CBDV (+287) from the other eluting cannabinoids. However, CBD (+315) and CBG (+317) co-elute.
Since reversed-phase was unsuccessful at separating CBG from CBD, I tried normal-phase. As it turns out, CBG is separable from both CBD and THC using this flash chromatography mode because of polarity differences, even at high loads (20% of silica mass), Figure 4.
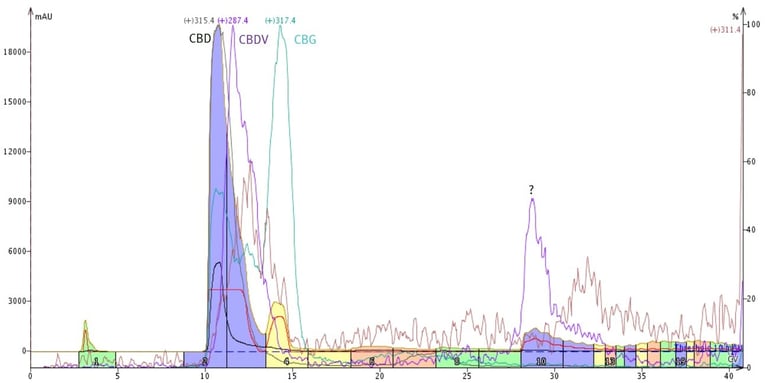
Figure 4. Successful purification of CBG from CBD and other cannabinoids in a hemp distillate using normal-phase flash chromatography.
I am sure by now that you have noticed the data in figures 1, 3, and 4 have numbers next to many of the “peaks”. These are the detected molecular ions of the various cannabinoids. In order to verify each UV-detected peak's identity, I used a flash chromatography system with an in-line mass detector (Biotage® Isolera Dalton 2000). This flash system simplifies flash chromatography by providing real time identification of an eluting compound’s molecular weight.
Using a flash system with an in-line mass detector is also useful when the compounds have the same molecular weight but are separated, e.g. CBD and THC, and when trying to determine the "unknowns", Figure 5.
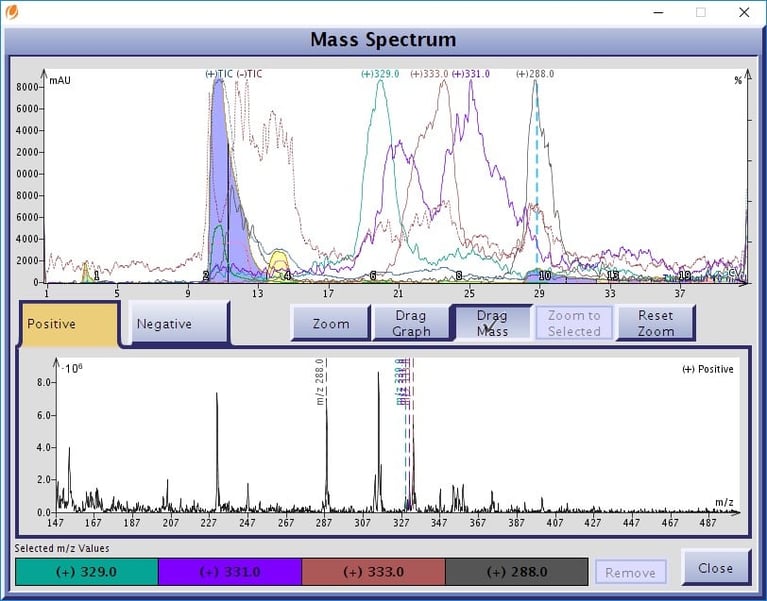
Figure 5. Flash chromatography with a mass detector enables identification and location of minor cannabinoids in an extract. In this hemp distillate we find four detectable masses, 329, 333, 331, and 288, which may be other cannabinoids.
These other detected compounds with masses 329, 331, 333, and 288 may also be cannabinoids. Possibilities include: cannabicoumarone (329), cannabielsoin (CBE) and/or CBDV-A (331), CBGV-A (333) or cannabichromaraone. The 288 mass does not match any cannabinoid or terpenoid that I know.
Interested in learning more about flash chromatography and cannabis? Check out our infographic available for download here:

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership