Cannabinoid isolation requires several steps from harvesting the biomass to extraction, winterization, decarboxylation (if desired), distillation, and finally chromatographic purification and isolation of the desired cannabinoids. It is during the chromatography step where water is introduced which, as we all know, can be challenging to remove from the desired cannabinoid by distillation or other evaporative technologies.
I have been asked by several of our customers on how best to remove water, which could be 30% of the fraction volume for CBD and potentially other cannabinoids. Currently used tools such as rotary evaporation are challenged when trying to evaporate water containing alcohol solutions.
Since cannabinoids are sticky, organic compounds that we know are bound by reversed-phase chromatographic media, I thought - why not use the same principle to remove water?
So, I decided to give it a try by first purifying a hemp distillate containing ~ 65% CBD by reversed-phase flash chromatography. For my research I prepared a sample of ~0.5 g of distillate in 2-mL methanol and then added 1.5 grams of loose C18 media (Biotage® KP-C18-HS) which I dried with a Biotage® V-10 Touch. This step allows dilute cannabinoid solutions to be concentrated prior to loading onto the purification column – it is not a required step for cannabis extract purification but more of a mechanism to load a larger than desired volume into the column.
I transferred the dried media to a dry load vessel (DLV) and attached it to the top of my equilibrated purification column (12-g Biotage® Sfär C18), Figure 1.

Figure 1. Hemp purification using a dry load vessel (DLV) attached to the main purification column.
The purification used a step gradient (80% methanol/20% water for 4 column volumes) followed by 100% methanol which provided a good separation. CBD was collected into fractions 1 and 2 (containing 20% water), while THC was collected in fractions 3 and 4 and CBCT in fraction 5 (all in 100% methanol), Figure 2.
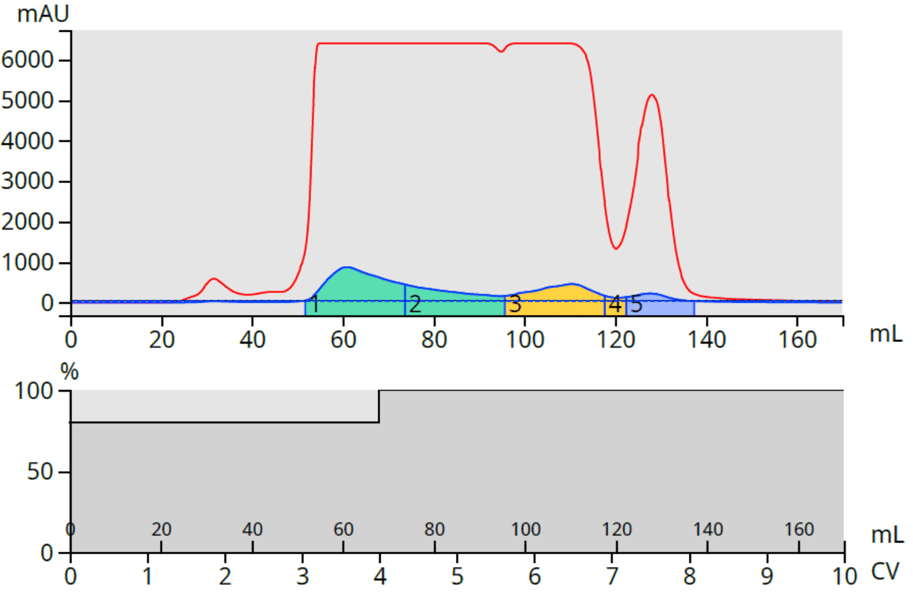
Figure 2. Hemp purification (0.5 gram) using a 12-gram Biotage® Sfär C18 column and dry load vessel (DLV). CBD was collected in fractions 1 and 2, THC in fraction 3 and CBCT in fraction 5.
This small-scale purification collected the CBD peak in 44 mL of 80:20 methanol. Larger scale purifications would of course generate much greater solvent volumes creating a water removal challenge and workflow bottleneck.
To determine my CBD fraction purity, I analyzed fractions 1 and 2 on a HPLC column and found them to be devoid of THC and other more lipophilic compounds, Figure 3.
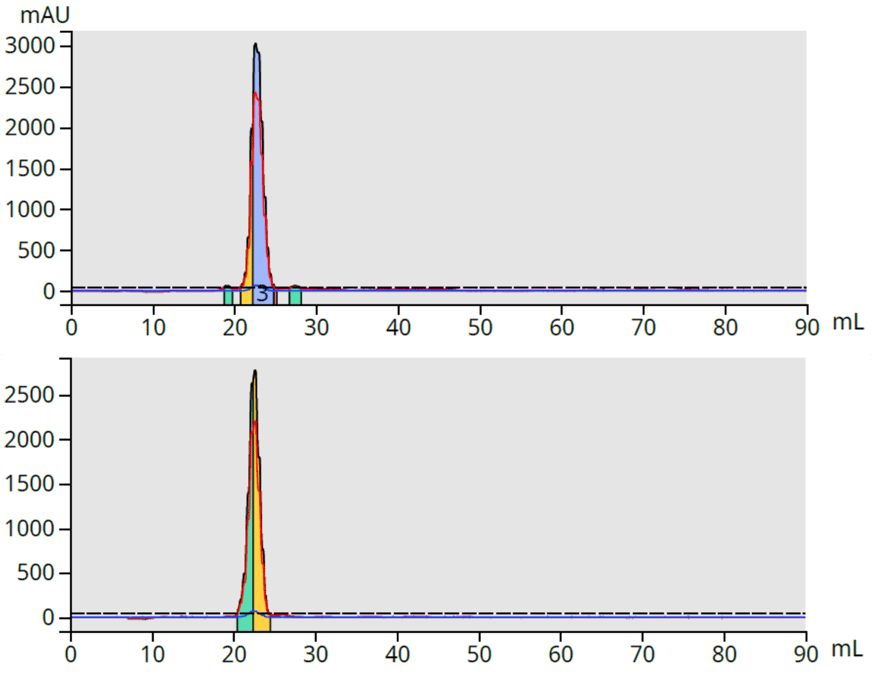
Figure 3. HPLC analysis of CBD fractions 1 and 2 showed both fractions to be THC-free.
To remove water from fractions 1 and 2, I added 1.4 grams of the same loose C18 media into a vial and added fractions 1 and 2, in ~5 mL aliquots, to the vial and evaporated using the Biotage V-10 Touch (36 °C, 30 mbar vacuum). I used only 1.4 grams of C18 because I wanted to maintain a ratio (1:3) of CBD to sorbent (an optimal ratio). Because the distillate was ~65% CBD, this should yield 0.325 g of purified CBD based on my 0.5 gram load.
The evaporation process was repeated until all of fraction 1 and fraction 2 were dried onto the C18 media. This technique evaporates the alcohol while adsorbing CBD and trapping the water in the media’s pores. For those unfamiliar with the Biotage V-10 Touch, it is designed for small volume evaporation. However, large scale purification fractions can be dried onto C18 media using rotary evaporation to achieve the same goal.
Once dried (yes, the result is a relatively dry but tacky material), I transferred the media into another DLV barrel and attached it to my Biotage® Selekt system, Figure 4.

Figure 4. DLV containing CBD impregnated C18 media mounted on the Biotage® Selekt purification system.
I then created the method below to elute CBD without water. For this method I disabled equilibration.
Elution method
Column: DLV
Solvent A: Water
Solvent B: Methanol
Equilibration: None
Gradient : 100% Methanol for 5 CV
Flow rate: 30 mL/min
Detection/collection parameters
λ- All (Collect): 198 - 400 nm
UV 1 (Show): 207 nm
UV 2 (Show): 275 nm
This method eluted CBD in 100% methanol which simplified its recovery, Figure 5.
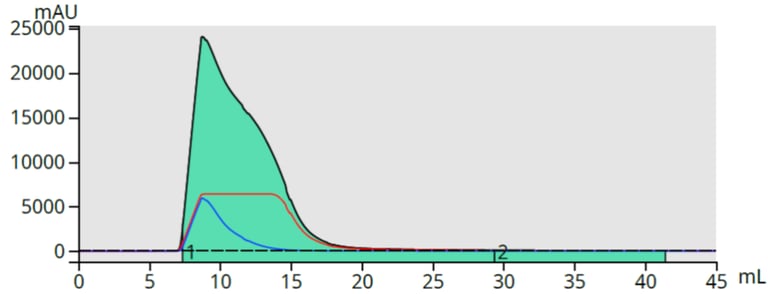
Figure 5. Water-free CBD fraction from DLV.
The collected CBD peak was dried and weighed 300 mg, indicating a 92% recovery from the initial purification (500 mg load, ~65% CBD = 325 mg of CBD potential).
This secondary purification step for CBD can be applied to any isolated cannabinoid in a partially aqueous solution and is an efficient method for removing water and improving cannabinoid recovery.
Want to learn more about cannabinoid purification? Please download our whitepaper Flash Chromatography Methods for Purifying Cannabinoids.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership