In recent years, most of the interest in the medicinal cannabis market has been focused on CBD (THC-free) and, to a lesser extent, THC. However, the market needs are changing with increasing interest now seen in the so-called minor cannabinoids. In this post, I will focus on cannabinol (CBN).
Since CBN is a degradation product of THC, it is naturally expressed in cannabis plants only in small quantities. Through oxidative processes (light/heat/air[1] or bio-enzymatically[2].), THC can be converted to CBN. Conversion of CBD to CBN is also possible using iodine (Federica Pollastro, 2018).
Whether through natural degradation/oxidation or by chemical/enzymatic reaction, there will be contamination from unreacted THC/CBD and other co-extracted compounds which need removal. But how do you purify the mixtures?
Crystallization is a possibility as pure CBN is a solid (Merck, 1989). However, it is my experience that flash chromatography is a more effective and efficient purification technique.
In most instances, reversed-phase flash chromatography is used to purify cannabinoids using relatively straightforward methods. However, as mentioned above, the reaction route from THC or CBD to CBN can generate multiple and different impurities, which can cause purification challenges as seen in, Figure 1.
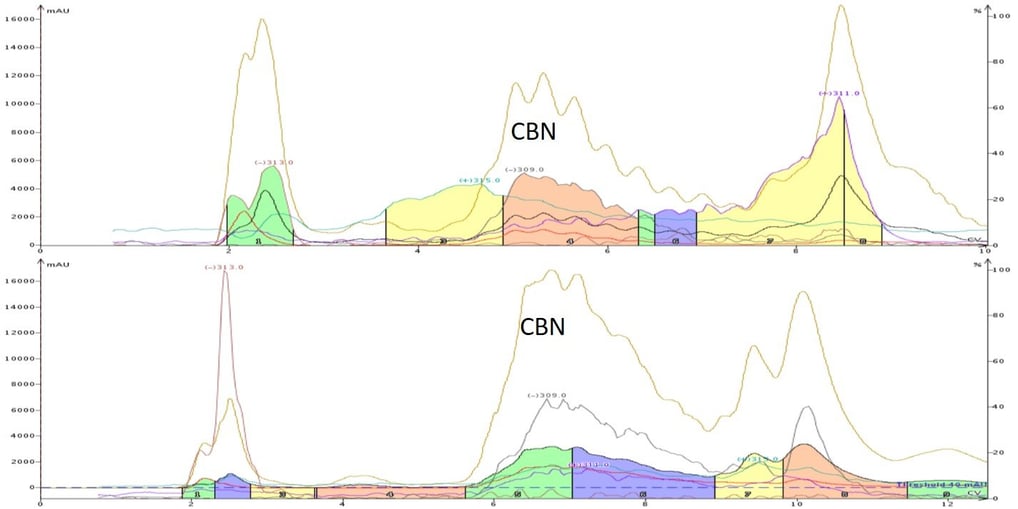
Figure 1. Crude CBN mixture purification comparison using reversed-phase flash chromatography with mass detection shows CBN (m/z -309) is separated from the other components. The top trace shows a crude CBN mixture with more contamination (yellow area in front of the CBN peak) than the mixture in the lower trace making the top sample more challenging to purify.
Depending on the raw material purity, reversed-phase may be all you need to go from a dark brown crude to clear, semi-crystalline CBN, Figure 2. The semi-crystalline CBN came from the evaporation of fractions 5 plus 6 from the lower trace in Figure 1.

Figure 2. Reversed-phase flash chromatography separated and purified CBN from a crude reaction mixture. Left - crude. Right - purified CBN.
But should your crude not be as easy to purify by reversed-phase, another flash chromatographic purification option is available, normal-phase. Normal-phase flash chromatography uses a silica column and organic solvents, typically hexane and ethyl acetate. This technique is required for CBG purification and, as it turns out, can be used for CBN purification (at least with the material in the top trace in Figure 1) as well.
So, how can you develop a suitable normal-phase flash method? I always begin with thin-layer chromatography (TLC) and evaluate different solvent combinations and ratios. For cannabis extracts, hexane with ether or hexane with ethyl acetate are useful. As ethyl acetate is a bit safer than ether (due to its higher boiling point, lower volatility, and higher flash point), I used it with hexane. Because these compounds are soluble in hexane, a very non-polar mobile phase is required. What I found was a TLC solvent blend of 3% ethyl acetate in hexane to provide a suitable separation of CBN from the other impurities, Figure 3.
The TLC data of my CBN crude reaction mixture, a hemp extract containing mostly CBD, and an aged cannabis extract of mostly THC, shows an elution order of CBD, THC, then CBN. Though the amount of separation isn’t great, it is enough to purify on a silica flash column using the same conditions.

Figure 3. TLC in 3% ethyl acetate/hexane solution of, from left, an aged cannabis extract (T), my CBN mixture (N), and a hemp extract (D). The data shows CDB eluting first followed by THC, and lastly CBN.
For flash chromatography, I set up my Isolera® Dalton 2000 system with a 10-gram Biotage® Sfär HC Duo silica column and programmed an isocratic method (3% ethyl acetate/97% hexane) with detection using mass settings m/z -309 and m/z -313 (negative ionization), m/z +311 and m/z +315 (positive ionization). I chose mass detection because I wanted to ensure I was detecting and collecting CBN, which has a different molecular mass (310) than CBD and THC, both of which have a mass of 314.
My results, using the more sensitive negative ionization mode, showed that this method successfully separated and collected CBN (detectable m/z -309) away from the other mass-detectable by-products (detectable m/z -313), Figure 4.
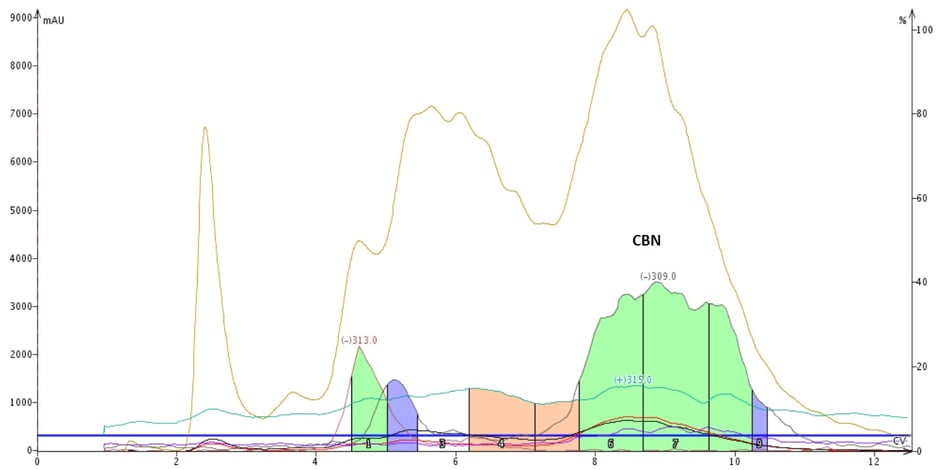
Figure 4. Normal-phase flash chromatography of my crude mixture separates CBN from the other impurities and by-products.
For comparison, I also used normal-phase flash chromatography using the same method for the aged cannabis and hemp extracts, Figure 5. The results follow the TLC results.
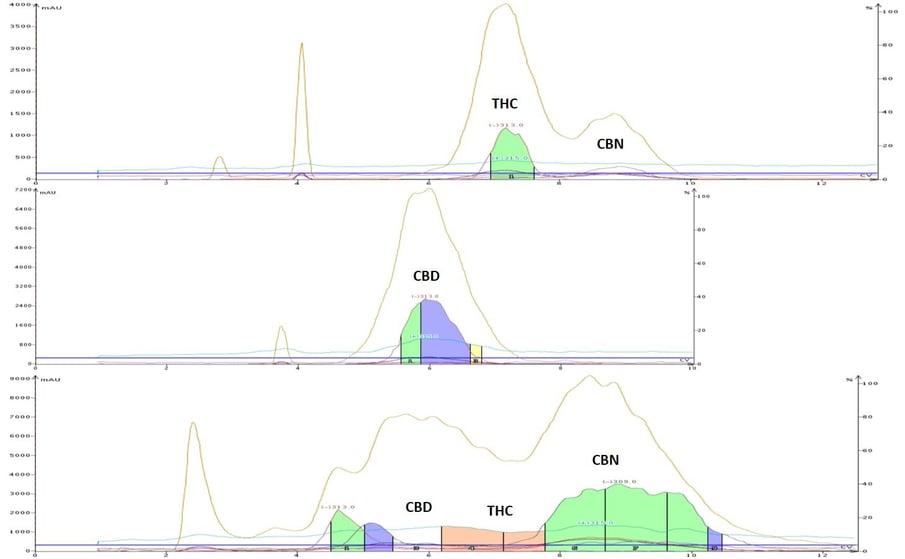
Figure 5. Normal-phase flash chromatography of Top - aged cannabis, Middle - hemp distillate, and Bottom - CBN crude mixture. This data shows excellent correlation to the TLC results. Graphs are normalized for run volume.
To verify my collected CBN purity, I pooled the CBN fractions, evaporated the solvent, re-dissolved in methanol, and ran my reversed-phase method. The result was a single peak with the correct CBN molecular mass, Figure 6.
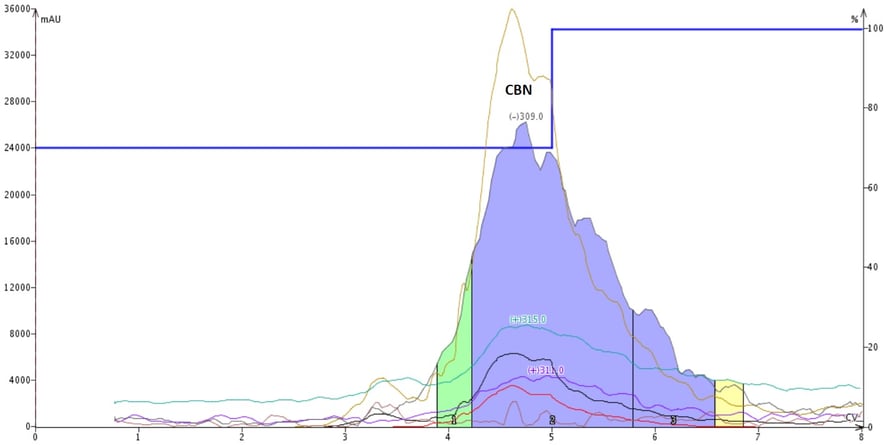
Figure 6. Reversed-phase flash chromatography of the isolated CBN fractions (via normal-phase) from Figure 4 show very high purity.
So, if you need to make and purify/isolate CBN, normal-phase flash chromatography is certainly a viable option if reversed-phase does not work.
For more information on flash chromatography, please download our whitepaper Flash Chromatography Methods for Isolating Cannabinoids.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership